Turning Up the Heat on Cancer
The fact that heat can help fight cancer has been known since antiquity: “Sometimes after a long period of high fever the cancer suddenly receded,” explains Caroline Maake, professor of anatomy at the University of Zurich. As we know today, this is because cancer cells are more sensitive to heat than healthy cells.
Extended and repeated exposure to high temperatures, or hyperthermia, results in the cells activating stress proteins, which tell the body’s immune cells to target the sick cells. Even only a minor temperature increase, for example to 43 to 45°C, is enough to kill off cancer cells. The elevated temperature spreads through the tumor and sensitizes the tissue so that it better absorbs drugs or radiation.
In other words, hyperthermia can improve the effects of chemo or radiation therapy. In Switzerland, hyperthermia is already being used in combination with standard treatment forms, but major clinical trials are yet to be carried out.
Tumors completely disappeared
Now Caroline Maake has teamed up with French physicist Edouard Alphandéry to study a novel approach for treating cancer with hyperthermia. The researchers are using magnetic nanoparticles containing iron oxide to treat clearly defined breast or brain tumors in mice.
These nanoparticles are injected directly into the tumors, a procedure that also reduces the risk of damaging healthy tissue. The particles are then activated from the outside by an alternating magnetic field, making their temperature – as well as the tumor’s – rise. The animal model has yielded some surprising findings: “Through hyperthermia alone, we made brain or breast tumors disappear completely within a month, without the mice showing any visible side effects,” explains Maake.
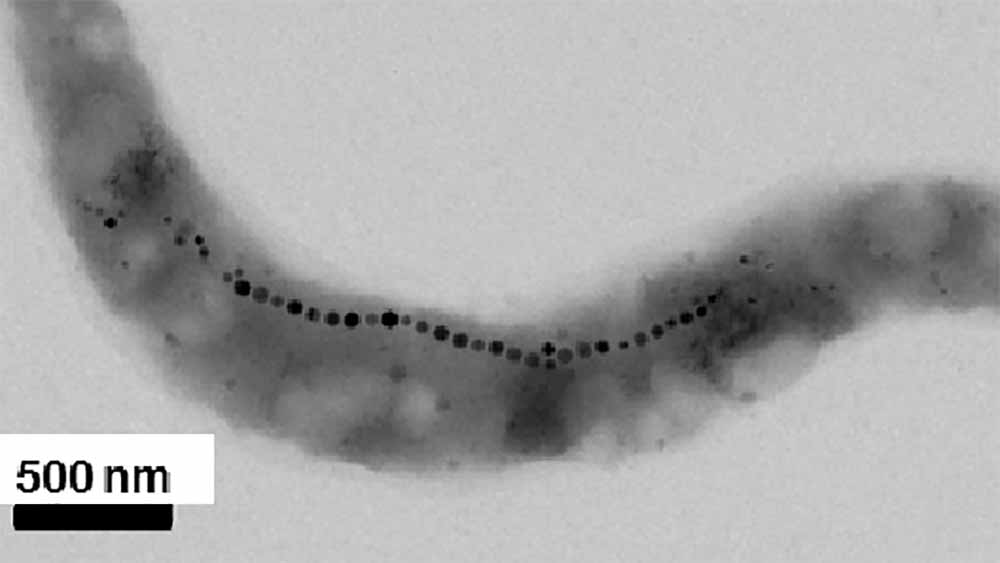
Nanoparticles with attractive properties
The magnetic nanoparticles used for the study are not produced in the lab, as is usually the case, but occur naturally. Edouard Alphandéry has developed a patented process for harvesting, and cleaning, the so-called magnetosomes from an aquatic bacterium. Caroline Maake is fascinated by these powerful little things: “They’re not only very effective but also beautiful.”
Under the microscope, magnetosomes are revealed to be chain-like structures on the inside of bacteria. Unlike artificially created magnetic nanoparticles, magnetosomes are uniform in size, heat-stable and highly pure – properties that make the new hyperthermia procedure particularly effective.
Treating malignant tumors in horses
Maake and Alphandéry have now been awarded funding to continue with their research into the benefits of this form of hyperthermia treatment. As part of the Eurostars framework program for research and innovation, the two scientists will work together with researchers at the Vetsuisse Faculty of UZH. Next spring, the UZH research group will start treating horses suffering from cancer.
The horses have malignant connective tissue tumors, often close to their eyes or mouths, which makes them extremely difficult to remove through surgery. Caroline Maake’s research project is no less difficult: “Translating our findings from mice to large animals is very challenging.”
One of the challenges involves getting the horses into an alternating magnetic field so that the nanoparticles can be stimulated. “This was relatively straightforward with mice, which we could easily put in a magnetic field. But this isn’t possible with horses, and this is why we’re testing alternative methods,” explains Maake.
Better use of immune response and imaging
The EU funding program isn’t the only support Caroline Maake’s research is getting: She’s also being backed by the NOMIS foundation to investigate further questions in connection with magnetosomes. Her goal is to find out what role the immune system plays when it comes to eliminating tumors: “We’ve established that nanoparticles are very effective, but we now want to know more about why this is the case. And we want to be able to make better use of possible immune responses,” says Maake.
Another aim is to improve compatibility with imaging techniques. Previous research demonstrates that magnetosomes can create powerful signals in the tumor, which can make it very difficult to get images of the cancer tissue and monitor the treatment progress. “We’re investigating how exactly the nanoparticles spread out and degrade over time. This will allow us to use just enough nanoparticles to kill the tumor without preventing us from getting images,” says Caroline Maake.