Predatory Bacteria
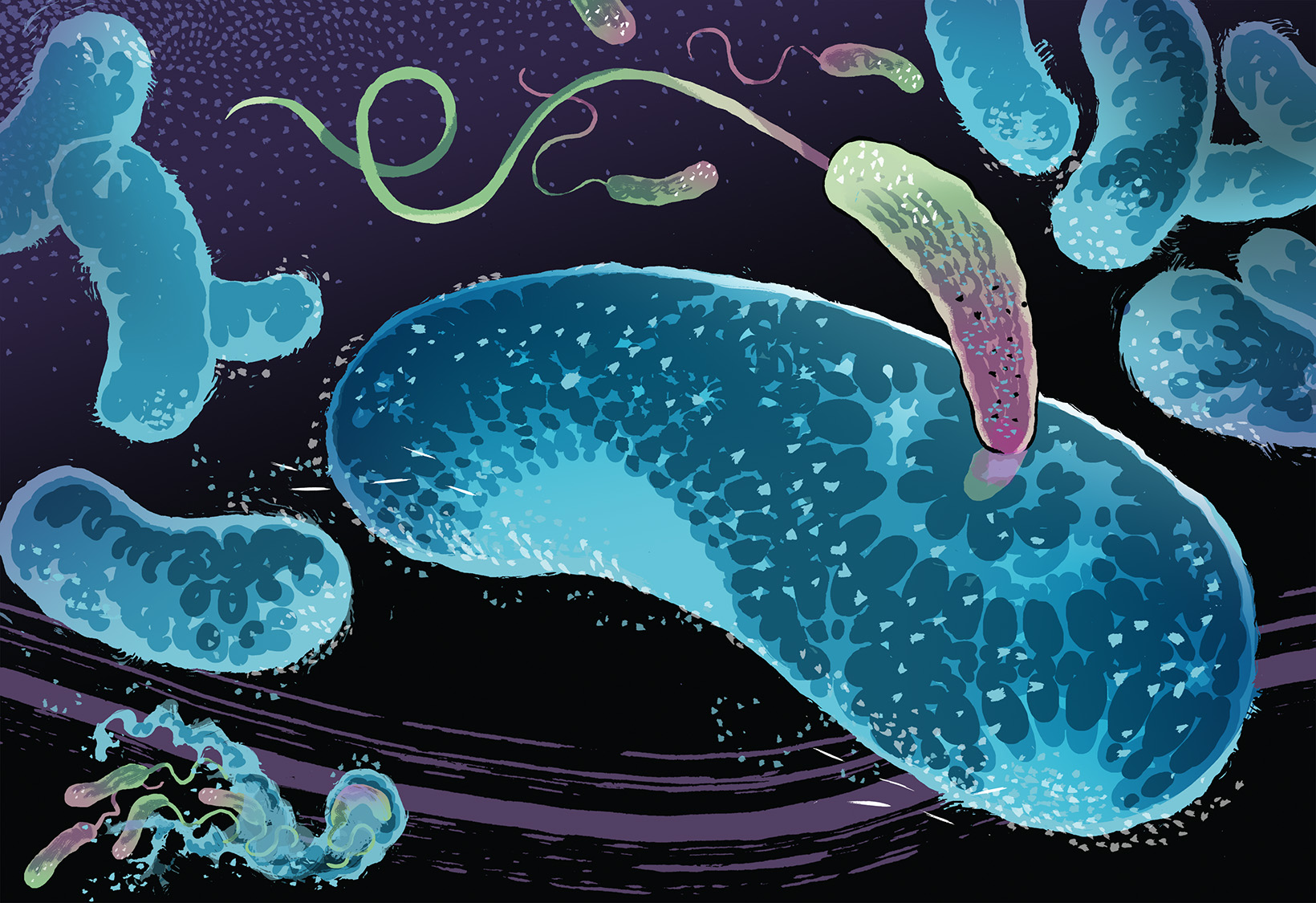
The spread of antibiotic-resistant bacteria is an enormous challenge for the healthcare system. It is almost impossible to stop the development of antimicrobial resistance, and there are almost no new kinds of antibiotics being developed that can be used against drug-resistant bacteria. Researchers are therefore looking to nature to find alternatives. They have set their sights on the natural enemies of pathogenic bacteria such as bacteriophages – viruses that kill bacteria – as well as predatory bacteria that attack, consume and eliminate other types of bacteria. Bdellovibrio bacteriovorus is one type of predatory bacteria that is of interest to researchers. It’s an ideal candidate because it likes to dine on gram-negative bacteria with antibiotic resistance. Experiments in the lab have shown that Bdellovibrio bacteriovorus is capable of killing and eating many kinds of pathogenic bacteria, including Escherichia coli, Enterobacter, Proteus, Serratia, Citrobacter, Yersinia, Shigellen, Salmonella and Vibrio. This varied meal plan is an advantage compared to bacteriophages, which usually prefer to eat one specific kind of bacteria.
Microbiologist Simona Huwiler is currently investigating the potential of B. bacteriovorus in a research project that is supported by the Fund for Promoting Young Academic Talent (FAN). Scientists have been aware of B. bacteriovorus for over 60 years, but the microorganism is receiving renewed attention due to the antibiotic crisis. Simona Huwiler first heard about predatory bacteria at a conference and then learned how to work with them in a laboratory setting during her postdoctoral studies at the University of Nottingham.
B. bacteriovorus is widespread and can be isolated from soil, wastewater and rivers. It is also a prime example of predatory bacteria and particularly suited for use in research. “B. bacteriovorus provides researchers with a kind of genetic toolkit for making changes to a bacterium,” explains Huwiler, whose research group works in Leo Eberl’s lab at the Department of Plant and Microbial Biology. What’s more, this strain is capable of killing various gram-negative bacteria that cause problems in hospitals. “There are concrete applications that make research very interesting,” says Huwiler.
Private dining room
Predatory bacteria attack and neutralize other types of bacteria. But what actually happens during one of these attacks? Using a microscope with 100x magnification, Simona Huwiler demonstrates in the lab how B. bacteriovorus attacks E. coli. First, she uses a pipette to transfer a laboratory strain of E. coli onto a glass slide. Then she adds a drop of liquid containing the much smaller predatory bacteria and puts the slide under the microscope. The view through the microscope reveals how the larger, rod-shaped E. coli float around sluggishly in the liquid while the much smaller predatory bacteria whiz around them at high speed, almost like annoying flies on a summer evening. After a few minutes, you can already observe how some of the predatory bacteria have docked onto an E. coli bacterium, similar to a ship that is being boarded. “Predator bacteria have a kind of stiff rope that they can use to temporarily attach themselves to potential prey. They then pull both the rope and themselves closer to the target,” says Huwiler.
In order to penetrate the prey, a predatory bacterium uses enzymes to bore a hole in the outer cell wall and then slips inside, sealing the hole behind it. This creates a kind of private dining room for the predator and prevents valuable nutrients from escaping. Once B. bacteriovorus is inside, the E. coli bacterium changes its shape, and within 20 minutes, it transforms from a long rod into a sphere. The predator grows inside of its dead prey until all of the nutrients have been consumed. It then begins to divide and multiply, and depending on the size of the prey, it can produce from two to ten offspring.
Well-planned escape
Once the prey has been fully consumed, the predator bacterium needs to reemerge from inside the cell wall. Huwiler researched this “escape phase” on a molecular level during her postdoc at the University of Nottingham, where she worked in the lab of Liz Sockett. “For me, this entry and exit phase is the most exciting part. It’s possible that the tools and mechanisms that the predator bacteria use to penetrate the cell wall could also be used as a type of new antibiotic,” says Huwiler.
During Huwiler’s postdoctoral fellowship, the team in Nottingham worked together with Andrew Lovering and was able to demonstrate that a specific enzyme, a modified lysozyme, is partially responsible for the ability of the predator bacteria to bore a hole in the cell walls of prey bacteria, allowing them to burst out again after feasting. “Predator bacteria probably use different methods to allow them to escape from inside the empty shells of their prey,” says Huwiler. These strategies – or other mechanisms, tools and weapons deployed by predatory bacteria – might be useful in the future as a new type of “medication” against infectious diseases.
An important question is whether prey bacteria are able to develop resistance against predatory bacteria like they do against antibiotics. It is precisely this question that Huwiler is researching in her current FAN-funded experiments, together with postdoctoral student Subham Mridha and biomedicine professor Rolf Kümmerli. Animal studies have shown that using B. bacteriovorus against disease-causing bacteria is well-tolerated. Huwiler is now investigating whether bacteria develop resistance to attack, a phenomenon that has not been very well documented in the literature. “From the perspective of evolutionary biology, one would expect resistance to develop in a race between predator and prey,” adds Huwiler. She says that it’s important for the experimental design to give the prey bacteria a chance to develop resistance.
Living antibiotics
In her experiments, Huwiler took eight different prey populations of E. coli, alternated exposing them to the predatory bacteria 15 times and then let them grow in a recovery phase in the absence of predators. In the following step, the genes of the prey population were sequenced before and after the experiment, as were the genes of the prey control group that was not exposed to any predators. The changes that occurred, including the possible development of resistance, are currently being investigated in greater detail. The experiments are expected to yield valuable information about the interaction between predator and prey bacteria. These insights are important for the future application of predatory bacteria as a living antibiotic or for their use as a treasure chest of possible new medications.