New Zurich-Made Microscope Proves Popular
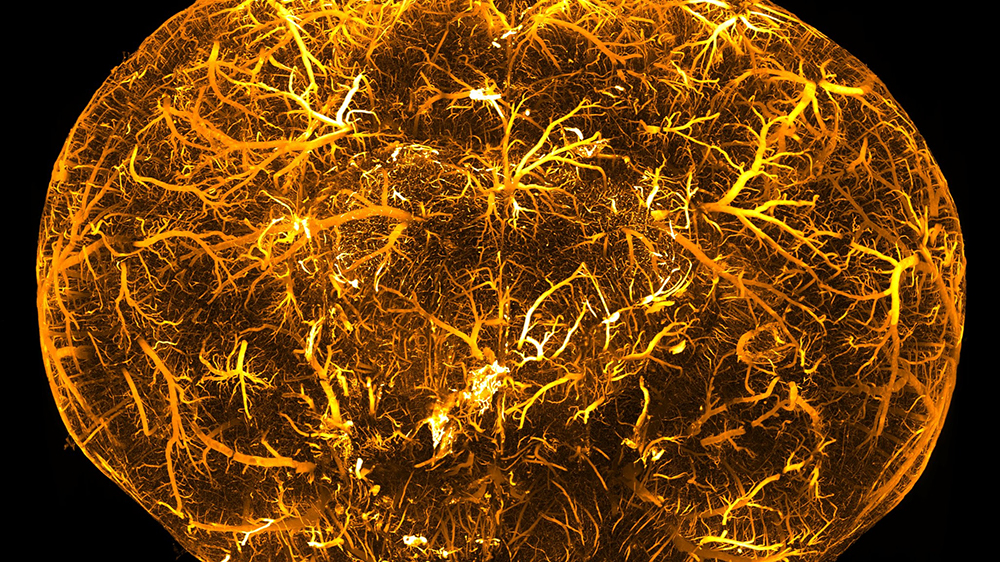
It all began with a query from the lab of Adriano Aguzzi: Some four years ago, the neuropathologist asked the team of neuroscientist Fritjof Helmchen whether it would be possible to microscope larger tissue samples using light sectioning. This method involves a focused laser beam scanning a plane of light through tissue that has been made transparent, enabling researchers to reconstruct structures within a sample in 3D, e.g. nerve cell networks or blood vessels.
PhD candidate Fabian Voigt, who was working on this microscopy technique in Helmchen’s lab, delved into the matter and started searching for a piece of equipment that met the required criteria: A device that could process samples the size of a finger within a matter of minutes, in sufficiently high resolution. However, none of the microscopes on the market met the needs of the researchers, especially with regard to processing time. So Voigt and his colleagues decided to build one themselves from individual parts that were available for sale.
Tricky configuration
“The main challenge was configuring the light beam, or light sheet, to fit the relatively large volume of the sample,” explains Voigt, who studied physics and neuroscience, and now works as a postdoc in the lab of Fritjof Helmchen. The laser scanning technology has to have the right intensity, speed and mode to produce an image in the desired resolution. Even the preparation of samples is a science unto itself. To prevent the light from bending and scattering, researchers first have to remove the lipids from the sample. Such clearing techniques have seen great progress over the past years and paved the way for the use of light-sheet microscopy in the life sciences. Last but not least, to be able to get an image at all, the target structures have to be tagged with fluorescent markers.
“Light sheet microscopy is practically an art form in itself,” says Voigt. It’s no surprise then that it took a while to properly assemble the components of the prototype at the Brain Research Institute and get the software and hardware to work. But he succeeded in the end, making it possible to capture entire mouse brains in only eight minutes using the “mesoscale selective plane illumination microscope”, or mesoSPIM. Voigt then worked together with Daniel Kirschbaum to build a second prototype in Adriano Aguzzi’s lab.
Demand from the community
The story of their success quickly spread across the scientific community, and soon external institutions came knocking at the doors of UZH. The first of these enquiries came from the Wyss Center at the University of Geneva and EPFL, which has since become the home of a third mesoSPIM. “That was the moment we decided to make the procedure’s details available on a website and publish an article on our work in a journal,” says Voigt.
The article in question is available in the prestigious Nature Methods journal, while the website mesospim.org documents the development, applications, technical details and, of course, images of the microscopes. The website also covers the specific details of the now seven devices currently in operation. Voigt is fascinated by the technology, which can “optically dissect” biological samples and display previously unknown structures within a matter of minutes. It is representative of the boom in microscopy techniques, which give researchers unprecedented access to the inner workings of biological systems.
Open science
But the mesoSPIM initiative is also an excellent example of independent cooperation among scientists, a concept often referred to as open science. “Scientific research can achieve more if we share techniques and data, and discuss our ideas with each other,” says Voigt.